
Committed to Excellence
Colorcon is committed to serving our customers through product and service excellence. By focusing on our customers' objectives, Colorcon develops solutions that align with your business strategies.
Reducing the Total Cost of Ownership is a key objective for many of our customers. Opadry® complete film coating systems deliver these results by eliminating the need for separate inventories of polymers, plasticizer, and pigment. Our customers can produce the highest quality product in the shortest time possible because Opadry systems provide batch-to-batch color and coating consistency. In addition, our plants, which operate to the same quality standards and manufacturing processes globally, are strategically located in key pharmaceutical markets allowing for superior on-time delivery service and a seamless business continuity plan.
Around the world, Colorcon has Continuous Process Improvement experts dedicated to customer focused process excellence. These lean program advocates can collaborate with you on robust product, service, and process improvements. If you have questions on Process Excellence or are interested in collaboration, contact us at ProcessExcellence@colorcon.com.
Top Tips for Flawless Film Coating
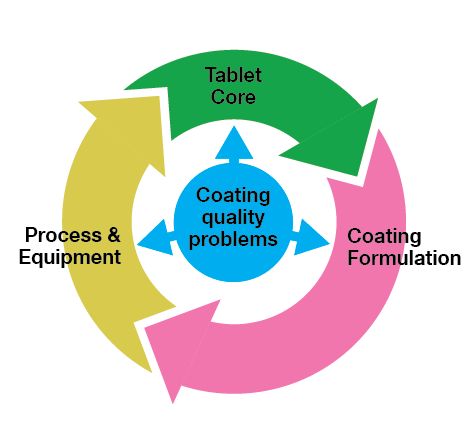
Need Additional Help to Perfect Your Film Coating Process?
Coating problems are usually the result of a combination of factors. Colorcon can help you identify common causes experienced during the coating process and give guidelines on how to resolve them quickly. Click here to access the Colorcon Guide to Troubleshooting.
Better Still - Expand Your Resources and Consult with the Experts.
Colorcon Technical Services are available to customers around the world to help shorten their development time.
We deliver solutions to our customers’ most critical product development needs. We can help you identify possible causes and suggest ways to resolve them, resulting in increased process efficiency and improved product quality.

Switch to PVA-Based Coating Helps Actavis, UK Improve Packaging Efficiency up to 75%
Situation
As a process improvement initiative, Actavis, a generic pharmaceutical manufacturer based in Barnstaple, UK, changed their coating formulation to drive efficiency for a large volume generic product. This was achieved by switching from an HPMC-based Opadry to a PVA-based Opadry II coating system where Actavis were able to simplify the packaging process by eliminating the addition of talc (a slip aid) resulting in improved packaging efficiency.
Coating Process Improvements
Comparative coating trials using Opadry (HPMC-based) and Opadry II (PVA-based) fully formulated systems were conducted using three different size oval-shaped tablets. The tablets were coated using a Glatt GC500 (19” Pan with 8 drive bars, modified baffles) and packed on a C90 blister packer. Commercial product coated with the HPMC-based Opadry and the additional talc step was used as control.
Results
Equivalent tablet weight gain and good color uniformity was easily achieved using a PVA-based Opadry II coating. The shape and size of the tablets demonstrated a difference in packaging speed, with the larger tablets showing an increase up to 75%. Removal of the talc step also eliminated dust generation normally observed in packaging with the current commercial batches.
Conclusion
Shape, size and film coating of tablets all impact packaging speed. The use of a PVA-based Opadry II coating enables faster packaging without the need for talc addition, resulting in faster, more cost effective process with reduced dusting.
| Packaging Speed (tablets/min) | ||||
| Tablet Size (mm) | Tablet Coating Color | Opadry HPMC With Talc | Opadry II PVA | Increase in Packaging Speed |
|---|---|---|---|---|
| 10.0 x 5.2 | Yellow | 190 | 200 | 5.3% |
| 12.7 x 6.5 | Orange | 210 | 250 | 19.0% | 14.5 x 7.5 | White | 200-220 | 350 | 59 - 75% |
Thanks to Actavis, UK for sharing this study.
Introducing Latest Innovation in Aqueous Enteric Coating
Many new chemical entities (NCE) present challenges to the formulator due to chemical instability in acidic media or they cause irritation to the gastric mucosa. The application of an easy-to-use, enteric coating can provide protection for the NCE and the patient.
Proton pump inhibitors (PPI) present a further challenge to the formulator as they require protection from the stomach acid, but also increase the pH of the stomach. In this case, an enteric coating that provides robust acid protection at elevated pH levels is required.
Acryl-EZE® II optimized aqueous acrylic enteric system is a fully formulated, easy-to-use coating that overcomes these challenges by offering protection at intermediate pH (up to 4.5), and subsequent rapid release in a higher pH (6.8) buffer media.
Colorcon recently presented two posters at the 2014 American Association of Pharmaceutical Scientists (AAPS) Annual Meeting that demonstrated intermediate pH protection on lansoprazole multiparticulates, as well as acetylsalicylic acid tablets coated at laboratory and pilot scale.

EUDRAGIT® is a registered trademark of Evonik Röhm GMBH, Dramstadt, Germany
A New High Performance Moisture Barrier Film Coating

High productivity immediate release (IR) film coatings with exceptional moisture barrier functionality, and no adverse impact on drug release, are increasingly required for pharmaceutical oral solid dosage forms.
Colorcon has developed a novel PEG-free IR film coating system, Opadry® amb II, High Performance Moisture Barrier Film Coating that exhibits excellent moisture barrier performance, high coating productivity and low impurity levels.
During development of Opadry amb II, Colorcon identified a new methodology to measure the moisture barrier performance of film coated tablets, and successfully used this to evaluate how coating formulation and coating thickness impact barrier performance of the film coated tablet. The results demonstrate the superior in-use functionality of this coating compared to other aqueous options.
AAPS Posters demonstrating the barrier properties of Opadry amb II and the new on-tablet testing methodology can be linked here.

Reduce Production Time and Cost
Using METHOCEL™ DC2
Through the global Controlled Release Alliance, Colorcon and Dow Pharma & Food Solutions together proudly announce the successful commercial launch of METHOCELTM DC2, premium cellulose ethers. This new, directly compressible HPMC, significantly reduces production time and costs in matrix tablet manufacture. Specifically engineered to provide more spherical particle morphology, the resulting superior flow properties of METHOCELTM DC2 successfully eliminate the need for costly and time-consuming wet granulation steps.
METHOCELTM DC2 delivers:
- Greater product consistency/uniformity, while reducing potential waste
- Shorter development time for quicker scale-up and streamlined manufacturing
- Lower manufacturing costs
- Elimination of wet granulation process (up to 60% cost savings)
- Convenient and reliable with low regulatory hurdles
Case studies demonstrating the consistency of API unit-dose uniformity from tablet-to-tablet and improved tablet performance are included in the webinar linked here.
Colorcon works together with Dow Pharma & Food Solutions through the Controlled Release Alliance to bring joint resources to accelerate pharmaceutical product development efforts for controlled release and reach markets throughout the world.
The Dow Diamond and METHOCEL/ETHOCEL/POLYOX are trademarks of The Dow Chemical Company.


A Strategy to Modulate Drug Release and Reduce Variability for Hydrophilic Matrix Tablets
In hydrophilic matrix tablets, low aqueous-solubility drugs pose challenges in modulation of release profiles and achieving consistent release in physiological conditions. To evaluate potential formulation strategies, Colorcon developed matrix tablets of hydrochlorothiazide using METHOCELTM premium cellulose ethers (K100 LV CR) and two fillers, lactose (soluble) or Starch 1500® partially pregelatinized starch (partially soluble). Then the matrix tablets were coated with Surelease®, ethylcellulose dispersion Type B NF, and a hydrophilic pore-former Opadry®, complete film coating system, at various ratios and weight gains.
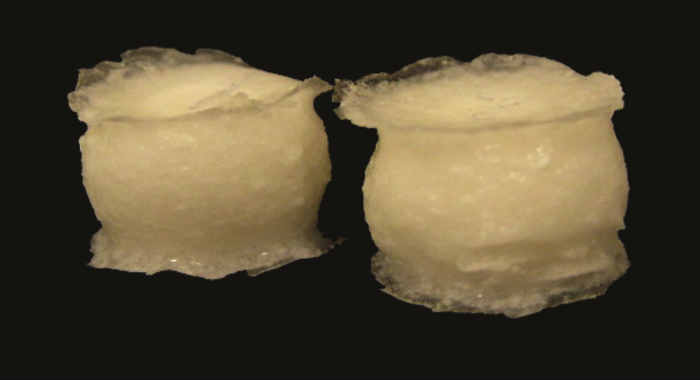
The results showed that drug release from uncoated matrix tablets was variable at different agitation rates during dissolution testing. An evaluation of tablets in bio-relevant media using physiologically relevant residence time indicated variable and higher initial release rate for uncoated matrices containing lactose but more robust behavior for tablets containing Starch 1500.
Dissolution profiles from the Surelease:Opadry coated tablets showed initial delay (lag time), followed by zero order release kinetics, with reduction or elimination of variability compared to uncoated matrices under different dissolution testing conditions. Such reduced variability may mitigate mechanical effects of post-prandial (fed) stomach. Effects of coating weight gain and inclusion levels of pore-former were critical in achieving robust and stable release profiles.
Article: Mehta RY, Missaghi S, Tiwari SB, Rajabi-Siahboomi AR, Application of Ethylcellulose Coating to Hydrophilic Matrices: A Strategy to Modulate Drug Release Profile and Reduce Drug Release Variability. AAPS PharmSciTech, May 2014. Read article »
Colorcon Survey Addresses Patient
Compliance & Safety Concerns
As color specialists and world leaders in film coating, Colorcon embraces its responsibility to help the pharmaceutical industry provide safe and effective medications for the patient. We recently carried out patient/consumer preference studies in various parts of the world to identify and understand the common challenges patients experience with recognizing and taking their medications. The results show that consumer concerns run from visual differentiation (recognition) through to difficulty in handling and swallowing.
Through the extended BEST® service, Colorcon helps customers select and modify tablet shape, size and color according to patient group and thus help address published FDA guidance by aiding compliance and reducing medication errors.

To read more, link here to the AAPS poster: Physical Appearance Preference for Solid Oral Dosage Formulations, or contact your Colorcon BEST representative.
Reduce Manufacturing Complexity
Create Economical, Low Friability Tablets Ready for Film Coating and Packaging
45 years of direct compression success with Starch 1500 is hard to overlook. The facts are clear, Starch 1500 when used as secondary excipient alongside microcrystalline cellulose (MCC), delivers tablet hardness and low friability, ideal for film coating and packaging.
Starch 1500, the only partially pregelatinized starch manufactured exclusively for the global pharmaceutical industry also provides the formulator:
- An economical disintegrant option for direct compression
- Stability for moisture sensitive drugs
- Effectiveness for low dose drugs
- Process flexibility for granulation
Link here to find out more.

Expanded Natural Colors - Blues, Greens and Purples

Colorcon is excited to begin offering fully formulated film coating and colorant systems using the natural blue pigment, Spirulina extract. With this addition to our broad range of non-synthetic pigment options we can now offer an expanded natural palette of blue, green and purple coating systems.
Currently, spirulina extract based film coatings are available for dietary supplement products in the EU and Canada, plus colorant systems for food and confections in the USA. Colorcon has submitted a color additive petition to the FDA to extend the regulatory acceptability of Spirulina extract for use in coatings for dietary supplement and pharmaceutical tablets and capsules in the US and completed extensive stability studies in support of this petition and the ongoing use of this pigment in our film coating systems.
As consumers of food and dietary supplement products increasingly look to avoid the use of artificial colorants, Colorcon is pleased to offer an ever-expanding selection of natural colorants for use in these applications.
To learn more, contact Colorcon.
Online MSDS Immediate Access Now Available

At Colorcon, we strive for continuous improvement to benefit of our customers. Accordingly, we recently added a powerful new MSDS Search function to our website which will allow visitors to immediately access MSDS in PDF format.
The new feature is accessible from the Regulatory and Compliance Tab, with an additional link from the site map on the website.
Simply type in the Colorcon product code, region (optional) and language (optional) to see a list of matching MDS documents in PDF format that can then be immediately downloaded. No password or login is required.
For direct access, link here.
Congratulations – IPEC Recognizes Colorcon CSO
Colorcon is proud to announce that Dr. Ali Rajabi-Siahboomi, VP and Chief Scientific Officer, was awarded the International Pharmaceutical Excipients Council (IPEC) Industry Research Achievement in Excipient Technology Award during the recent American Association of Pharmaceutical Scientists (AAPS) Annual Meeting, San Diego, California. The IPEC Awards were established to encourage the growth of technology related to excipients for the betterment of the public and medical science.


Colorcon Safety Studies Support Higher Levels of PVAP or PVAP-T than Previously Used in Approved Drugs
Pharmaceutical companies conducting material safety reviews on the use of polyvinyl acetate phthalate (PVAP) or co-processed polyvinyl acetate phthalate-TiO2 (PVAP-T) can now access two articles in the public domain for use in their regulatory filings related to recent safety studies conducted by Colorcon.
One article published in October 2014 reported the maternal and developmental no-observable-adverse-effect level (NOAEL) of PVAP was the highest concentration administered, i.e., 3.0% (equivalent to 2324 mg PVAP/kg/day).1 Another article published in August 2014 summarizes 90-day and genotoxicity studies that were also performed.
This data, along with the developmental study, was reviewed by the IPEC New Excipient Safety Evaluation Panel, which concluded that the safety data supports usage levels in solid oral dosage forms up to 829 mg of PVAP or PVAP-T per day.2
PubMed Abstract links for each article are provided below:
Oral dietary developmental toxicity study with polyvinyl acetate phthalate (PVAP) in the rat Read artcle
Safety of PVAP and PVAP-T including a 90-day dietary toxicity study in rats and genotoxicity tests with polyvinyl acetate phthalate (PVAP) Read article
Copies of the referenced articles are available through Colorcon’s Online Request system.
1. DeMerlis C.C., Schoneker D.R., Borzelleca J.F., Oral dietary developmental toxicity study with polyvinyl acetate phthalate (PVAP) in the rat, Regulatory Toxicology and Pharmacology October, 2014.
2. DeMerlis C.C., Schoneker D.R., Borzelleca J.F., Safety of PVAP and PVAP-T including a 90-day dietary toxicity study in rats and genotoxicity tests with polyvinyl acetate phthalate (PVAP), Food and Chemical Toxicology , August, 2014 C.C. DeMerlis, D.R. Schoneker and J.F. Borzelleca



