Protecting the Supply Chain
Reports continue of threats to the Pharmaceutical industry supply chain, with numerous well-documented incidents due to misrepresented, counterfeit or adulterated materials in medicinal products. Some of these have even led to deaths.
Additionally, the continuity of a safe supply can be threatened by many types of risks. Geographic, climatic or weather related changes can, for example, impact crop yields, damage manufacturing plants or interrupt transportation networks; while political change, acts of terrorism or industrial disputes may also result in a negative impact.
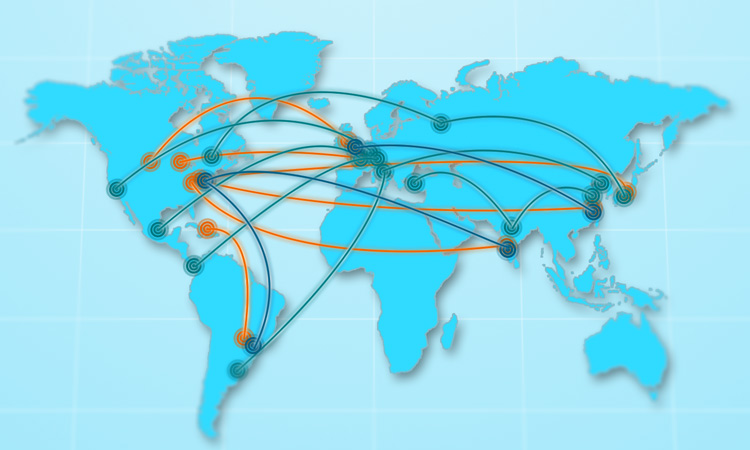
Colorcon's Global Network
Regulators around the world are now focusing more on excipients and starting materials. The finished dosage form manufacturer is required to have a thorough knowledge of this supply chain, and some new legislative requirements are now in place to strengthen this.
At Colorcon we understand the importance of managing risk in the excipient supply chain and the potential impact this may have on your final product.
With a thorough knowledge and assessment of all the individual materials we use at Colorcon, our customers, in turn, can have confidence that they will fulfill their regulatory obligations.
Through our global manufacturing and distribution network, Colorcon manages potential risk to our customers and helps maintain continuity of your production, protecting the supply chain and ultimately ensuring patients get their medicines.
Business Continuity Planning requires the management and mitigation of the threats to the wider supply chain. To learn about Colorcon’s Plan click here
 Technical
TechnicalMoisture Protection beyond the Package
Defend your product stability from the inside out
For moisture-sensitive actives, protection from environmental elements like humidity is a necessity. Unwanted exposure to moisture can impact the stability of these products. A common strategy is to use high barrier packaging (which can be costly) and to utilize low moisture content excipients within the tablet core.
In a recent article published in Pharmaceutical Manufacturing and Packaging Sourcer, Elizabeth Shen, Ph.D., discusses the significance of film coating and excipient selection when designing moisture sensitive dosage forms. The article features a case study using Opadry® amb II, fully formulated aqueous moisture barrier coating, to preserve the stability of amoxicillin and clavulanic acid in real-time and accelerated stability studies. The study showed that tablets coated with a traditional hypromellose (HPMC) based coating showed significant degradation, while tablets coated with Opadry amb II showed little to no decline in drug assay after six months accelerated stability, regardless of packaging. Insight into the excipient selection, water activity, and moisture content is also presented.
Announcing the Colorcon Academy™
At Colorcon we are always looking to do something extra. This year, with the introduction of the Colorcon Academy, we’ve connected our highly valued education programs continuing to advance our global standard in education.

Since the late 1960’s, with our very first Colorcon Coating School®, to today, we have expanded our training programs to offer continuing education to thousands around the world. We continue to strengthen our commitment to deliver value beyond expectations.
Under the new Colorcon Academy platform, Colorcon will offer five different programs at all of our 20 technical services facilities worldwide, in addition to programs designed by our customers held at their site. Attendees at Academy events continue to advance their skills in pharmaceutical sciences and become champions in their organizations.
Our goal is to address current industry challenges and ensure our customers are equipped with the very best knowledge and practical expertise to positively impact their organization.
To read more about the variety of programs offered this year, Click here »
Increased Monograph Compliance - Starch 1500®

Colorcon is pleased to announce the immediate availability of a global grade of Starch 1500®, partially pregelatinized maize starch. The new grade meets the wider monograph compliance to NF, EP, JPE and the recently created CHP requirements, plus the Indian Pharmacopeia regulations. Colorcon worked diligently to establish this new grade to help our customers’ source a single tableting excipient that meets the broadest range of regulatory requirements.
Starch 1500® manufactured exclusively for the pharmaceutical industry in dedicated GMP facilities. The process results in partial pregelatinization and increased particle size, with improved flow properties and compatibility for use in solid dose formulations compared to alternative starches. It is particularly useful when used for low dose applications and moisture sensitive actives.
In an environment of increased scrutiny on the purity and reliability of pharmaceutical excipient supply by regulatory bodies around the world, Colorcon’s new global grade of Starch 1500® is your choice when wider monograph compliance is a must for your organization.
Bitter to Better
Taste masking of bitter APIs is required to produce palatable formulations, especially for pediatric products. Functional coatings are one option that can be applied to a variety of substrates having differing physiochemical properties. While these functional coating options do exist, selection of the ideal functional coating formulation requires careful consideration.
Recent collaboration studies between Colorcon and Merck were aimed to help identify key bio/food-relevant evaluations that would inform pharmaceutical scientists of coatings that will be suitable. Results of these studies are summarized in the joint posters between Colorcon and Merck at the Annual AAPS Conference, 2016.
In the first study, a coating combination of Surelease® and Opadry® was used to evaluate taste-masking a soluble and bitter tasting API. Surelease, containing ethylcellulose, creates the barrier, while the Opadry (HPMC-based) is the “pore-former” assisting in dissolution. The study results demonstrate how formulation, process and characterization techniques that mimic and forecast in vivo performance can guide the development of palatable formulations to improve the patient experience.
Skip the Yuck: Guiding development of taste-masking formulations in pharmaceutical development

The second study, to identify ideal process parameters, was completed using a selected pH-insensitive coating formulation on mini-tablets. Observations of physical changes in the morphology and phase behavior of the coating were made using scanning electron microscopy (SEM), atomic force microscopy (AFM), and focused ion beam milling – Helium ion microscopy (FIB-HIM).
Retest Dates for POLYOX™ Water Soluble Resins Now Extended
You asked. We responded.

The industry has been requesting longer retest intervals for POLYOX™, to enable wider use of this polymer for modified release applications.
You will be happy to hear that retest intervals for all the mid-molecular weight grades of POLYOX™ in 140-lb drums are being extended from 6 months to 12 months, including LEO, non-LEO, FP, and SFP grades. Colorcon, through the Controlled Release Alliance, supplies POLYOX™ based on analytical testing to ensure that material is within the viscosity specification. Dow now confirms the material may be used beyond the retest date if appropriate viscosity testing and examination confirms it is still suitable for use. This allows you to use POLYOX™ confidently with reduced frequency of retest.
 Regulatory & Quality
Regulatory & QualityStreamlining the Drug Approval Process
Colorcon provides information and resources that will help streamline the regulatory approval process for all customers using our products.
Through My Colorcon™, customers can request regulatory and quality information directly through our website 24/7. Many documents are automatically delivered within minutes of the request, with improved accuracy and rapid response.
Our Global Regulatory Affairs Department has devised Regulatory Filing packages for key market areas. They contain information needed by regulators to accurately assess the quality and safety of our products in that region.
Colorcon is also establishing eDMFs for certain products to satisfy the requirements of Health Canada and the U.S. FDA and can provide electronic Letters of Authorization (LOAs) to customers to reference these eDMFs, where needed. In most cases, however, all the information for appropriate regulatory assessment can be supplied directly to customers through the Colorcon online request system, providing a confidentiality agreement is in place.
Colorcon will continue to develop new systems and documents in the future to further assist customers using our products in streamlining the drug approval process for their drug products.
 News
NewsColorcon China Answers Generic Industry Challenges

Colorcon China Team
China's leading exhibition for the pharmaceutical manufacturing sector, API China, represents raw materials, intermediates, ingredients, processing and packaging machinery. Colorcon China has continued to participate in the API exhibition for the past 12 years. This event provides exciting opportunities for both business and networking with customers and colleagues from across the country.
An important topic this year for generic manufacturers, is the drug re-evaluation requirement, bringing the challenge to demonstrate equivalency with the originator. Colorcon’s regulatory knowledge and global experience of solid oral dose bring unmatched technical service and support to navigate through these requirements successfully.
As a company delivering innovative products and exceptional services in the region, we are always excited to showcase “What’s New.” This year, we launched Opadry® QX, a unique coating to improve process efficiency across a wide range of coating equipment, while delivering the perfectly elegant finish. Opadry QX is a revolutionary product, with 40% reduction in coating time, significantly improving production capacity and lower overall cost of production.





