
Colorcon Recognizes
30 Years in Japan
Colorcon is delighted to recognize the 30th anniversary of business activities in Japan. In September 1986, Colorcon (Japan) Limited opened the first office in Nihonbashi, Tokyo, followed in 1991 with the first Colorcon Technical Services Laboratory established in Itabashi-ku, Tokyo. This early investment brought specialist technical support and speedy availability of custom-formulated film coating samples directly to customers, with local coating trials and formulation services, plus regulatory expertise to support Japanese companies directly.
To meet the growing demand for Colorcon film coating products to be produced directly in Japan, further investment was made in a local manufacturing facility located in Shibakawa-cho, Fujinomiya. The new facility including the relocation of our technical and sales offices, opened in March 2000. The facility received the industry recognized full Pharmaceutical GMP Manufacturing License, demonstrating Colorcon products meet the stringent demands of the pharmaceutical industry; a production to supply our Japanese customers was set to begin.
Since 2003, additional capacity has been added to support the growing market demand and the Pharmaceutical GMP Manufacturing License continues to be renewed every 5 years, demonstrating our ongoing commitment to delivering the highest quality product and maintaining the trust of our customers.
Contributing to continuing education, the inaugural Colorcon Coating School™ educational event was conducted in the early 1990s, providing pharmaceutical professionals with a deeper knowledge and practical experience for development and production for oral solid dosage forms. Colorcon also maintains a close association with academia and sponsors the prestigious Colorcon Award, presented annually at the “Symposium on Particulate Preparation and Designs”, organized by The Society of Powder Technology Japan.

Thomas Pilgram, General Manager, Colorcon Japan, extends his personal thanks to our valued customers for their continued business, and reveals a special 30th anniversary event, the Colorcon Innovation Program, to take place on 14th October 2016 at the Conrad Hotel Tokyo.
 The Innovation Program will include the latest advances and technical applications in the areas of film coating, core formulation and modified drug release for solid dosage forms. Colorcon is honored to announce that Professor Hirofumi Takeuchi from Gifu Pharmaceutical University will be a keynote speaker at this event, alongside Dr Ali Rajabi-Siahboomi, Vice President and Chief Scientific Officer who will attend the forum and present on behalf of Colorcon Inc.
The Innovation Program will include the latest advances and technical applications in the areas of film coating, core formulation and modified drug release for solid dosage forms. Colorcon is honored to announce that Professor Hirofumi Takeuchi from Gifu Pharmaceutical University will be a keynote speaker at this event, alongside Dr Ali Rajabi-Siahboomi, Vice President and Chief Scientific Officer who will attend the forum and present on behalf of Colorcon Inc.
LINK TO REGISTER »
or call Colorcon directly Tel+81-544-65-2711

Pharmaceutical Packaging
Colorcon’s No-Tox® Printing Inks – Making Products Safe Inside
When manufacturing solid oral dosage drugs, the potential of contaminating products during production is a key concern. Precautions to prevent this, such as clean-room environments, special equipment, protective clothing and strict cleaning procedures for employees entering the manufacturing space help mitigate the risk.
The same precautionary mindset must apply when packaging the manufactured product. All aspects of the packaging materials, including the printing inks used for blister packs, bottle seals, inserts and desiccants require careful consideration to keep the product safe inside the final package.
Colorcon’s No-Tox inks use only FDA 21 CFR compliant ingredients. They are designed exclusively for printing applications involving direct and indirect contact with pharmaceutical, food, and medical products. Most importantly, all Colorcon No-Tox inks are manufactured under cGMP processes and come with a batch-specific written 21CFR compliance guarantee.
Colorcon No-Tox® Pharmaceutical FDA Compliant Printing Inks offer a wide variety of options for pharmaceutical packaging.
Colorcon Hits the Target on Productivity
& Risk Reduction
Colorcon is excited to share recent updates on how we are helping our customers increase their productivity and reduce regulatory risk at the key pharmaceutical events happening during the remainder of this year.
Opadry® QX, a quick and fleXible tablet film coating, launched in April 2016, is rapidly growing in demand. As a revolutionary advance, this new immediate release film coating significantly reduces preparation and production times, ensuring coating process efficiency and lower manufacturing costs across various types of equipment. It can be applied over a broad range of process conditions and always results in a premium quality finish for tablets, even those containing temperature-sensitive active ingredients. Multinational pharmaceutical companies can easily transition the manufacture of oral dosage forms across continents; overcoming challenges often met with equipment differences.
There’s also breaking news, including the opening in January 2017 of Colorcon’s 7th film coating manufacturing in Indaiatuba, Brazil and the expansions and process integration of our Suglets® sugar sphere plants in Stoughton, Wisconsin and Bazainville, France. We’re making investments to manage supply chain continuity and further strengthen our global business continuity plan, reducing risk for our valued customers.

We look forward to meeting with you.
- AAPS – Denver, Colorado – 13-17 November, Booth #1215
- CPHI – Mumbai, India - 21 - 23 November, Stand #D13 (BEC)
- API - Shanghai, China – 16-18 November, Stand A4E88
When it comes to Film Coating,
There’s more than meets the Eye
In the commentary, Formulating for the Perfect Tablet Finish, Ali discusses the importance of selecting the right system, and how getting it ‘wrong’ can impact patient compliance, or even compromise drug efficacy by negatively affecting the release profile. So, when choosing, it’s vital to select the best solution and make sure consideration is given to factors such as taste-masking, ease of swallowing, desired release profile, regulations for the intended market, plus the overall aesthetic appearance. From the manufacturer’s perspective, coated tablets facilitate branding and market differentiation, as well as offering the potential to incorporate functional attributes for dose delivery. For the patient, it’s all about compliance to achieve the correct therapeutic dose and helping to reduce the risk of medication errors.
With expert insight, you’re much more likely to get your coating first time right.
In a recently published article in The Medicine Maker, Dr. Ali Rajabi-Siabhoomi, Vice President and Chief Scientific Officer at Colorcon shares his insight on choosing the right coating solution and keeping up with trends.

Stability & Consistency = Reliability You Can Trust
Concentrations of the release controlling components of a coating, such as film forming polymers and pore-formers, determine the extended release performance of barrier membrane coated multiparticulates. Ensuring the compositional stability of coating formulations is key to achieving consistent performance over time.
In support of the product launch of Opadry® EC, Ethylcellulose Organic Coating System, Colorcon presented a study at the 2016 Annual Meeting of the Controlled Release Society. The poster “Compositional and Performance Stability of a Fully Formulated Ethylcellulose Barrier Membrane Coating System” results show the effect of different concentrations of polymer, pore-former and plasticizer for Opadry EC formulations over time and indicated compositional stability following six months accelerated storage stability testing.
The release profiles of drug-layered coated multiparticulates were shown to be consistent, regardless of powder storage condition or storage duration. Finally, consistent drug release was obtained from Opadry EC coated multiparticulates stored under intermediate and accelerated storage conditions for six months.

Dose-Weight Proportional Extended Release Formulations

Developing a modified release formulation for a multiple strength active pharmaceutical ingredient (API) is challenging and can be time-consuming. After optimizing for one target quality product profile (one dosage strength), the next lower or higher dosage strength comes with its own development issues. If the formulation has unique customization, it may mean different qualitative or quantitative changes, different stability tests, scale-up, and validation for each strength. A dose-weight proportional formulation can greatly simplify the process.
Colorcon recently presented a poster entitled “Study of Dose-Weight Proportionality in Extended-Release Multiparticulate Systems” at 2016 Controlled Release Society Annual Meeting held in Seattle, Washington, USA. The purpose of this work was to formulate dose-weight proportional extended release multiparticulate formulations of a model drug at different doses, using drug layered and barrier membrane coated sugar spheres. When developing a dose-weight proportional multiparticulate formulation, using similar batch of multiparticulate with optimal release profile, surface area to volume ratio and surface area to film thickness ratio remains constant for different multiple strengths.
This work was a continuation of evaluation of different sustained release formulations strategies to achieve dose-weight proportionality, such as osmotic push-pull tablet formulations and hydrophilic matrix tablet formulations.
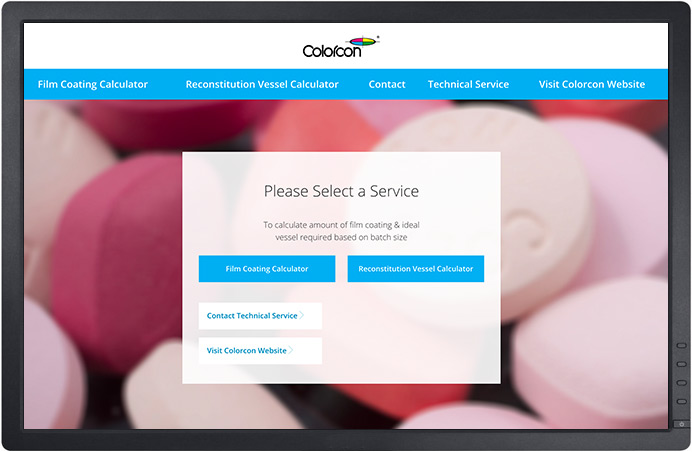
New On-Line Tool – Colorcon Coating Calculator
To support the use of Colorcon’s fully formulated coating systems, a new on-line Coating Calculator App is now available to registered customers.
This simple-to-use tool calculates the amount of Colorcon film coating required and recommends the ideal dispersion vessel dimensions, based on batch size, for your Colorcon coating system.
Whether at development or production scale, this tool will help you achieve the perfect finish.
Adopt a Safety by Design Approach for NDA, ANDA & OTC Applications
In April 2016, the US Center for Drug Evaluation and Research (CDER) published a new Guidance for manufacturers “Safety Considerations for Product Design to Minimize Medication Errors in Drug Product Development” to help reduce the potential for medication errors. The guidance encourages the use of size, shape, and color for the benefit of prescribers and patients to easily identify their medications.
Colorcon’s Technical Paper “Safety by Design” offers a concise summary of the key points covered in this Guidance. Moreover, our Brand Enhancement (BEST) team is ready to provide guidance and materials that will help support your NDA, ANDA or OTC submission to ease its way through the application process. Read the summary and let us know how we can help.


Colorcon and BASF Advance Co-operation
Following the press announcement on May 25th 2016, Colorcon is now pleased to confirm the final closure of transactions to acquire Kollicoat® IR Coating Systems from BASF. Colorcon will now serve all existing customers of Kollicoat® IR Coating Systems from its worldwide production facilities.
The addition of the Kollicoat® IR Coating Systems complements Colorcon’s existing film coating product line, reinforces our position as a leading innovator of fully formulated film coating systems, and provides customers with a wide range of film coating options.
To learn more please contact Colorcon Customer Care or your local representative.

Dow and Colorcon Extend and Broaden the Scope of the Controlled Release Alliance
Effective January 1, 2017, Colorcon Inc. and The Dow Chemical Company will broaden their existing Controlled Release Alliance to include Dow’s advanced technology based pharmaceutical excipients. Press Release »

Congratulations – APSGB Recognizes Colorcon Chief Scientific Officer
The Academy of Pharmaceutical Society of Great Britain (APSGB) recently recognized Dr. Ali Rajabi-Siahboomi, Vice President and Chief Scientific Officer at Colorcon with their Eminent Fellowship Award.
“The Fellowships are awarded based on significant contribution in the field of pharmaceutical science and Eminent Fellows provide the APSGB Board with advice and guidance on topics within their area of expertise.”
Colorcon congratulates Ali on this wonderful achievement.

When manufacturing solid oral dosage drugs, the potential of contaminating products during production is a key concern. Precautions to prevent this, such as clean-room environments, special equipment, protective clothing and strict cleaning procedures for employees entering the manufacturing space help mitigate the risk.
The same precautionary mindset must apply when packaging the manufactured product. All aspects of the packaging materials, including the printing inks used for blister packs, bottle seals, inserts and desiccants require careful consideration to keep the product safe inside the final package.
Colorcon’s No-Tox inks use only FDA 21 CFR compliant ingredients. They are designed exclusively for printing applications involving direct and indirect contact with pharmaceutical, food, and medical products. Most importantly, all Colorcon No-Tox inks are manufactured under cGMP processes and come with a batch-specific written 21CFR compliance guarantee.
Colorcon No-Tox® Pharmaceutical FDA Compliant Printing Inks offer a wide variety of options for pharmaceutical packaging.

& Risk Reduction
Colorcon is excited to share recent updates on how we are helping our customers increase their productivity and reduce regulatory risk at the key pharmaceutical events happening during the remainder of this year.
Opadry® QX, a quick and fleXible tablet film coating, launched in April 2016, is rapidly growing in demand. As a revolutionary advance, this new immediate release film coating significantly reduces preparation and production times, ensuring coating process efficiency and lower manufacturing costs across various types of equipment. It can be applied over a broad range of process conditions and always results in a premium quality finish for tablets, even those containing temperature-sensitive active ingredients. Multinational pharmaceutical companies can easily transition the manufacture of oral dosage forms across continents; overcoming challenges often met with equipment differences.
There’s also breaking news, including the opening in January 2017 of Colorcon’s 7th film coating manufacturing in Indaiatuba, Brazil and the expansions and process integration of our Suglets® sugar sphere plants in Stoughton, Wisconsin and Bazainville, France. We’re making investments to manage supply chain continuity and further strengthen our global business continuity plan, reducing risk for our valued customers.

We look forward to meeting with you.
- AAPS – Denver, Colorado – 13-17 November, Booth #1215
- CPHI – Mumbai, India - 21 - 23 November, Stand #D13 (BEC)
- API - Shanghai, China – 16-18 November, Stand A4E88
There’s more than meets the Eye
In the commentary, Formulating for the Perfect Tablet Finish, Ali discusses the importance of selecting the right system, and how getting it ‘wrong’ can impact patient compliance, or even compromise drug efficacy by negatively affecting the release profile. So, when choosing, it’s vital to select the best solution and make sure consideration is given to factors such as taste-masking, ease of swallowing, desired release profile, regulations for the intended market, plus the overall aesthetic appearance. From the manufacturer’s perspective, coated tablets facilitate branding and market differentiation, as well as offering the potential to incorporate functional attributes for dose delivery. For the patient, it’s all about compliance to achieve the correct therapeutic dose and helping to reduce the risk of medication errors.
With expert insight, you’re much more likely to get your coating first time right.
In a recently published article in The Medicine Maker, Dr. Ali Rajabi-Siabhoomi, Vice President and Chief Scientific Officer at Colorcon shares his insight on choosing the right coating solution and keeping up with trends.

Concentrations of the release controlling components of a coating, such as film forming polymers and pore-formers, determine the extended release performance of barrier membrane coated multiparticulates. Ensuring the compositional stability of coating formulations is key to achieving consistent performance over time.
In support of the product launch of Opadry® EC, Ethylcellulose Organic Coating System, Colorcon presented a study at the 2016 Annual Meeting of the Controlled Release Society. The poster “Compositional and Performance Stability of a Fully Formulated Ethylcellulose Barrier Membrane Coating System” results show the effect of different concentrations of polymer, pore-former and plasticizer for Opadry EC formulations over time and indicated compositional stability following six months accelerated storage stability testing.
The release profiles of drug-layered coated multiparticulates were shown to be consistent, regardless of powder storage condition or storage duration. Finally, consistent drug release was obtained from Opadry EC coated multiparticulates stored under intermediate and accelerated storage conditions for six months.

Dose-Weight Proportional Extended Release Formulations

Developing a modified release formulation for a multiple strength active pharmaceutical ingredient (API) is challenging and can be time-consuming. After optimizing for one target quality product profile (one dosage strength), the next lower or higher dosage strength comes with its own development issues. If the formulation has unique customization, it may mean different qualitative or quantitative changes, different stability tests, scale-up, and validation for each strength. A dose-weight proportional formulation can greatly simplify the process.
Colorcon recently presented a poster entitled “Study of Dose-Weight Proportionality in Extended-Release Multiparticulate Systems” at 2016 Controlled Release Society Annual Meeting held in Seattle, Washington, USA. The purpose of this work was to formulate dose-weight proportional extended release multiparticulate formulations of a model drug at different doses, using drug layered and barrier membrane coated sugar spheres. When developing a dose-weight proportional multiparticulate formulation, using similar batch of multiparticulate with optimal release profile, surface area to volume ratio and surface area to film thickness ratio remains constant for different multiple strengths.
This work was a continuation of evaluation of different sustained release formulations strategies to achieve dose-weight proportionality, such as osmotic push-pull tablet formulations and hydrophilic matrix tablet formulations.
New On-Line Tool – Colorcon Coating Calculator
To support the use of Colorcon’s fully formulated coating systems, a new on-line Coating Calculator App is now available to registered customers.
This simple-to-use tool calculates the amount of Colorcon film coating required and recommends the ideal dispersion vessel dimensions, based on batch size, for your Colorcon coating system.
Whether at development or production scale, this tool will help you achieve the perfect finish.

Adopt a Safety by Design Approach for NDA, ANDA & OTC Applications
In April 2016, the US Center for Drug Evaluation and Research (CDER) published a new Guidance for manufacturers “Safety Considerations for Product Design to Minimize Medication Errors in Drug Product Development” to help reduce the potential for medication errors. The guidance encourages the use of size, shape, and color for the benefit of prescribers and patients to easily identify their medications.
Colorcon’s Technical Paper “Safety by Design” offers a concise summary of the key points covered in this Guidance. Moreover, our Brand Enhancement (BEST) team is ready to provide guidance and materials that will help support your NDA, ANDA or OTC submission to ease its way through the application process. Read the summary and let us know how we can help.

Following the press announcement on May 25th 2016, Colorcon is now pleased to confirm the final closure of transactions to acquire Kollicoat® IR Coating Systems from BASF. Colorcon will now serve all existing customers of Kollicoat® IR Coating Systems from its worldwide production facilities.
The addition of the Kollicoat® IR Coating Systems complements Colorcon’s existing film coating product line, reinforces our position as a leading innovator of fully formulated film coating systems, and provides customers with a wide range of film coating options.
To learn more please contact Colorcon Customer Care or your local representative.

Effective January 1, 2017, Colorcon Inc. and The Dow Chemical Company will broaden their existing Controlled Release Alliance to include Dow’s advanced technology based pharmaceutical excipients. Press Release »

Congratulations – APSGB Recognizes Colorcon Chief Scientific Officer
The Academy of Pharmaceutical Society of Great Britain (APSGB) recently recognized Dr. Ali Rajabi-Siahboomi, Vice President and Chief Scientific Officer at Colorcon with their Eminent Fellowship Award.
“The Fellowships are awarded based on significant contribution in the field of pharmaceutical science and Eminent Fellows provide the APSGB Board with advice and guidance on topics within their area of expertise.”
Colorcon congratulates Ali on this wonderful achievement.



